
Are you worried about the risk of fire to your products or property? A small accident can quickly turn into a devastating event, but many materials are dangerously flammable without special treatment.
A flame retardant is a substance added to materials to prevent or slow the spread of fire. It is a critical safety measure that interferes with the combustion process, absorbing heat and isolating the material from the flames.

From my perspective at our aluminum hydroxide plant, I see flame retardant1s as a form of insurance built directly into a product. We produce a simple white powder, but when it’s added to plastics or textiles, it becomes a silent guardian. The concept sounds straightforward, but how these materials achieve this protection is a mix of simple physics and clever chemistry. Understanding this is key to choosing the right solution, so let’s look closer.
What do you mean by flame retardant?
The term "flame retardant" is everywhere, but what does it really guarantee? This lack of clarity can cause confusion about a material’s actual safety performance in a fire.
By "flame retardant," we mean a functional additive that makes a material more difficult to ignite and slows the rate of burning. It doesn’t make things fireproof, but it dramatically reduces flammability and gives people more time to escape.

The most important goal is increasing safety. A flame retardant’s job is to interfere with what is known as the "fire triangle2"—fuel, heat, and oxygen. If you can remove any one of these three elements, the fire goes out. Flame retardants are designed to attack one or more of these components during a fire. It’s not about making a material invincible. It’s about buying precious time.
The Goal is Escape Time
The key objective is to delay "flashover3." This is the point in a fire where everything in a room bursts into flames at the same time because of the intense radiant heat. By slowing the initial fire on one object, flame retardants can prevent or significantly delay flashover. This gives occupants critical seconds or minutes to evacuate and gives first responders a much better chance of controlling the incident. I always tell my customers that the aluminum hydroxide we supply for their products isn’t just a chemical; it’s a life-saving feature that protects people when things go wrong.
What is fire retardant made of?
You know they are important, but what are these protective materials made from? With growing concerns about chemical safety, choosing the right source is more important than ever.
Flame retardants are made from a diverse group of chemicals. The main families are mineral-based compounds like aluminum hydroxide, halogenated compounds containing bromine, and others based on phosphorus or nitrogen.

In my work, I focus on mineral flame retardants4 because they are seen as one of the safest options. Our main product, aluminum hydroxide (ATH), is essentially a purified mineral. It’s a fine white powder that is physically mixed into a material, like plastic, without chemically becoming part of the plastic’s own structure. This is different from other types that react with the material.
A Comparison of Common Types
The trend in the industry is moving toward halogen-free options due to environmental and health regulations. This is where materials like ATH shine. As a direct producer, we can ensure the purity and particle size meet the exact needs for making a safe, effective, and compliant final product. The table below breaks down the main options.
| Retardant Type | Main Component | How it Works | Key Advantage |
|---|---|---|---|
| Mineral | Aluminum Hydroxide (ATH) | Releases water, cools, and dilutes gas | Halogen-free, low smoke, non-toxic |
| Halogenated | Bromine, Chlorine compounds | Interrupts combustion in the gas | Very effective at low doses |
| Phosphorus | Various phosphorus chemicals | Forms a protective char layer | Works well in plastics and foams |
| Nitrogen | Melamine and other N-compounds | Releases inert gas, promotes charring | Often used with phosphorus retardants |
What are examples of flame retardants?
All the chemical names can sound abstract and confusing. It’s much easier to understand their importance when you see where they are used in products around you.
Common examples include Aluminum Hydroxide (ATH), which is widely used in the insulation for electrical cables. Brominated flame retardants (BFRs) are often found in the plastic casings of electronics like TVs and computers.

Seeing our product used in a final application is always fascinating. One of our biggest markets is for wire and cable manufacturers. They buy our aluminum hydroxide powder and mix it directly into the PVC or rubber that insulates the copper wires. A simple electrical short circuit could easily start a fire, but because our ATH is in the insulation, the fire is suppressed immediately at the source. It prevents a small problem from becoming a catastrophe. This single application protects homes, offices, and industrial facilities all over the world.
Safety in Everyday Objects
Beyond cables, our ATH and other flame retardants are used in many places:
- Construction: In wall panels, roofing membranes, and flooring to meet strict building safety codes.
- Transportation: In seats, carpets, and plastics for cars, trains, and airplanes to slow fire spread and give passengers time to exit.
- Electronics: While BFRs have been common, there is a strong push to replace them with halogen-free options5 like ATH in plastic housings and circuit boards. This is driven by buyers who want safer, more environmentally friendly products.
How do flame retardants work?
We know they stop fire, but how do they actually do it? The science seems like it would be very complicated, but it’s based on very direct principles.
Flame retardants work through physical or chemical actions. Some cool the material by releasing water vapor. Others form a solid insulating char layer. A third type releases chemicals that disrupt the chain reaction of fire in the gas phase.
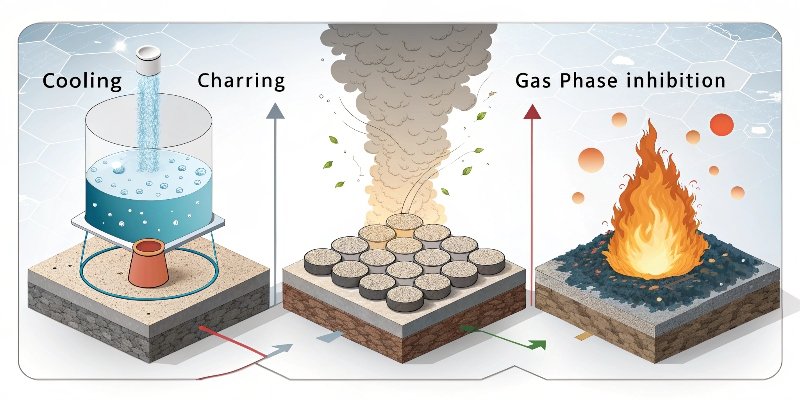
This is my favorite part to explain, because our product, aluminum hydroxide, has a particularly elegant and effective way of working. It uses a physical mechanism that is simple, powerful, and safe. It’s not a complex chemical trick; it’s a basic endothermic reaction6.
Cooling, Diluting, and Shielding
When a material containing aluminum hydroxide (ATH) is exposed to fire, it starts to break down at around 220°C. The chemical reaction is simple: it decomposes into aluminum oxide and water vapor. This process does three things at once to fight the fire:
- Cooling: The reaction is endothermic, meaning it absorbs a large amount of heat energy. This cools the plastic or rubber, keeping it below the temperature it needs to keep burning. It acts like a built-in heat sink.
- Dilution: The reaction releases a lot of water vapor. This water vapor dilutes the flammable gases that the burning material produces, making the air-fuel mixture too lean to ignite easily.
- Shielding: The aluminum oxide left behind is a non-combustible ceramic material. It forms a protective char layer on the surface of the material, acting as a shield that insulates the underlying material from heat and oxygen.
Conclusion
A flame retardant is a critical safety additive. It works by cooling, isolating, or disrupting combustion to slow or stop the spread of fire, ultimately saving lives and property.
-
Understanding flame retardants is crucial for safety in various materials and products. ↩
-
The fire triangle is essential for understanding fire dynamics and how to combat them. ↩
-
Flashover is a critical concept in fire safety; knowing it can save lives during emergencies. ↩
-
Mineral flame retardants like aluminum hydroxide are considered safer; learn why. ↩
-
Halogen-free options are safer for health and the environment; explore their benefits. ↩
-
Endothermic reactions are key to flame retardant function; understand their role. ↩
You may also be interested in:

What protective measures should be taken when handling aluminum hydroxide powder in a factory?
Are you worried about worker safety when handling fine powders? Aluminum hydroxide is generally safe, but mishandling its dust can lead to serious respiratory issues and even create explosive conditions.

What Key Technical Indicators Should Be Considered When Purchasing Aluminum Hydroxide?
Are you struggling to find the right aluminum hydroxide for your needs? You might be paying for features you don’t even require. Let’s simplify the technical details. When buying aluminum

What are the market trends for aluminum hydroxide in 2026?
Are you planning your 2026 raw material purchases? Volatile markets make it hard. I will share insights from the factory floor to help you make better decisions for your business.

Is aluminum hydroxide toxic and is it harmful to the human body?
Worried about the word "aluminum" in your products? It sounds scary and can make you question its safety. I’m here to clear up the confusion with simple facts. Aluminum hydroxide

What are the side effects of long-term use of stomach medications containing aluminum hydroxide?
Struggling with constant heartburn? Reaching for that stomach medicine might seem like the only option. But what if that relief comes with hidden long-term costs? The most common side effect

Does ingesting aluminum (such as through aluminum hydroxide) cause Alzheimer's disease?
Are you worried that everyday products containing aluminum might be harmful? This fear connects to serious health concerns like Alzheimer’s, making you question what is safe. Let’s look closely. Based

Why do some vaccines contain aluminum hydroxide (aluminum adjuvant)?
Confused about vaccine ingredients? Seeing aluminum hydroxide listed can be unsettling. I’ll explain its crucial role and why it’s there to help your body build strong immunity. Aluminum hydroxide acts

Is the aluminum adjuvant in vaccines safe for infants or adults?
You read about vaccine ingredients and worry. The word "aluminum" sounds scary, especially when talking about babies. But understanding the facts can give you peace of mind. Yes, the aluminum

What environmental problems are generated during the production of aluminum hydroxide?
Worried about environmental compliance in your supply chain? Sourcing from China can be complex. You need a reliable, eco-conscious partner for your aluminum hydroxide needs. The main environmental issue is

Where are China's main aluminum hydroxide production areas?
Are you struggling to find the right aluminum hydroxide supplier in China? This confusion can lead to higher costs and unstable quality, which hurts your business and your reputation. China’s