
Confused by chemical terms that sound alike? Assuming an "antacid" is just any "base" is a simple mistake that can lead to dangerous misunderstandings about safety and use.
An antacid is a type of base, but not all bases are antacids. An antacid is specifically a weak base that is safe for human consumption to neutralize stomach acid, whereas most other bases are highly corrosive and unsafe.

This distinction is more than just words; it’s a matter of safety and function. In my factory, we produce aluminum hydroxide1, a chemical that is technically a base. However, it only becomes a safe and effective antacid2 after it goes through a strict purification process3 to meet pharmaceutical standards. For my clients, like Mr. Park who supplies Korean pharmaceutical companies, understanding this difference between a general chemical and a specialized medical ingredient is the most important part of their job. Let’s break it down.
Is an antacid the same as a base?
Using chemical terms interchangeably feels efficient, but it’s a risky habit. Calling any base an antacid could lead to a serious error in sourcing or formulation. Let’s fix that.
No, they are not exactly the same. An antacid is always a base, but a base is not always an antacid. "Base" is a very broad chemical category, while "antacid" is a specific job description for a weak, medically safe base.

Think of it like this: all cars are vehicles, but not all vehicles are cars. A "base" is like the broad category of "vehicle," which includes everything from bicycles to cargo ships. An "antacid" is like the specific category of "car"—it has a very particular function and set of safety features. Many bases are powerful, corrosive industrial chemicals. For example, sodium hydroxide (lye) is a very strong base used as a drain cleaner. Ingesting it would cause severe internal damage. An antacid, on the other hand, must be a base weak enough and pure enough to react gently with stomach acid without harming the delicate lining of the esophagus and stomach. It’s a specialist chosen for a sensitive job.
Industrial Base vs. Antacid Base
| Feature | Industrial Base (e.g., Sodium Hydroxide) | Antacid Base (e.g., Aluminum Hydroxide) |
|---|---|---|
| Strength | Very Strong / Corrosive | Weak |
| Primary Use | Manufacturing, Cleaning | Neutralizing Stomach Acid |
| Safety | Highly Dangerous to Ingest | Safe to Ingest in Recommended Doses |
| Purity Level | Industrial Grade | High-Purity Pharmaceutical Grade |
What are the differences between antacids?
You’re staring at a pharmacy shelf with dozens of antacid options. They all promise relief from heartburn, but how are they different, and which one is right for you?
The main differences are in their active chemical compound. This determines their speed of relief, how long the effect lasts, and what side effects they might have, such as constipation, gas, or a laxative effect.

Formulating an antacid is a balancing act. Every active ingredient has pros and cons, which is why there are so many different products.
- Calcium Carbonate (e.g., Tums®): This is a very fast and powerful acid neutralizer. However, the chemical reaction produces carbon dioxide gas, which can lead to bloating and belching.
- Aluminum Hydroxide (e.g., in Maalox®, Mylanta®): This is the product we make. It’s slower to start working but provides longer-lasting relief. Its main side effect is that it can be constipating.
- Magnesium Hydroxide4 (e.g., Milk of Magnesia): This one works moderately fast, but it’s famous for its laxative effect.
To create a better product, manufacturers often combine ingredients. This is why you’ll see aluminum hydroxide and magnesium hydroxide mixed together in a single product. The constipating effect of the aluminum is balanced by the laxative effect of the magnesium. It’s a clever formulation strategy to give the user effective relief without a strong side effect in either direction.
Antacid Comparison
| Active Ingredient | Speed | Duration | Common Side Effect(s) |
|---|---|---|---|
| Aluminum Hydroxide | Slower | Long | Constipation |
| Magnesium Hydroxide | Moderate | Moderate | Laxative Effect |
| Calcium Carbonate | Fast | Short | Gas, Bloating |
| Sodium Bicarbonate | Very Fast | Very Short | High Sodium, Gas |
What is the difference between acid and antacid?
The words "acid" and "antacid" sound related, and they are. But not knowing the exact relationship means you’re missing the key to how these common medicines actually work.
An acid and an antacid are chemical opposites. An acid, like the hydrochloric acid in your stomach, has a low pH value. An antacid is a base with a higher pH value, designed to react with and neutralize the acid.

The best way to think about this is using the pH scale5, which measures how acidic or basic a substance is. The scale runs from 0 to 14.
- Anything below 7 is an acid. Your stomach acid is very strong, with a pH of around 1.5 to 3.5.
- A value of 7 is neutral, like pure water.
- Anything above 7 is a base (or alkaline). An antacid is a weak base.
When you have heartburn6, it’s because that strong stomach acid is in the wrong place. The job of the antacid is to perform a neutralization reaction. It’s a chemical reaction where a base cancels out an acid. For my product, the reaction is: Al(OH)₃ (the antacid) + 3HCl (stomach acid) → AlCl₃ + 3H₂O. The end products are aluminum chloride and plain water, which are much more neutral and don’t cause that painful burning sensation. It’s a simple and elegant solution to a very common problem.
What makes a base different from an acid?
You’ve heard the terms "acid" and "base" since high school science class. But what is the fundamental chemical difference that makes them opposites? The answer is surprisingly simple.
At its core, an acid is a substance that gives away a proton (a hydrogen ion, H⁺), while a base is a substance that accepts a proton. This simple act of giving or taking a proton defines their entire chemical character.
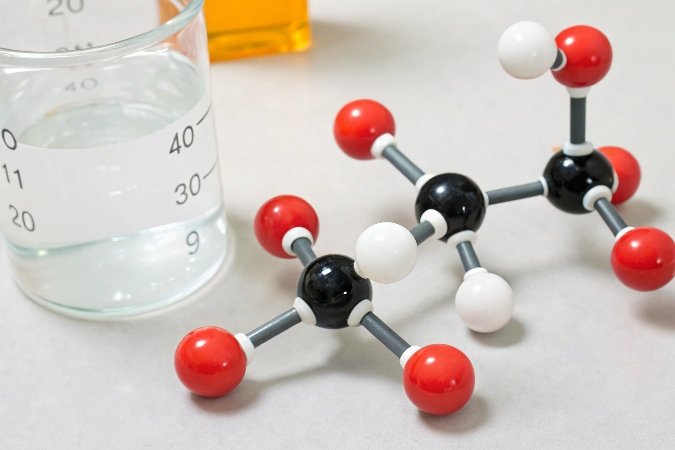
Let’s visualize this. An acid, like hydrochloric acid (HCl), is eager to get rid of its hydrogen proton (H⁺) when in water. This release of protons is what makes the solution acidic. Now, introduce a base like aluminum hydroxide, Al(OH)₃. The hydroxide parts (OH⁻) of the base are excellent at grabbing onto those free-floating protons. When an OH⁻ accepts an H⁺, they combine to form H₂O—a perfectly neutral water molecule. By accepting and locking up the protons that the acid released, the base effectively cancels out the acid’s effect.
This proton-exchange relationship is the foundation of their opposition. One is a donor, the other is an acceptor. This is why throwing a base into an acid is the perfect way to neutralize it.
Acid vs. Base: The Key Differences
| Feature | Acid (e.g., Stomach Acid) | Base (e.g., Antacid) |
|---|---|---|
| Role with Protons | Donates Protons (H⁺) | Accepts Protons (H⁺) |
| Effect on pH | Lowers the pH (makes it < 7) | Raises the pH (makes it > 7) |
| Simple Example | Lemon Juice | Soap or Baking Soda |
| Chemical Interaction | Is neutralized by bases | Neutralizes acids |
Conclusion
An antacid is simply a specialized, weak base that is safe to eat. It works because it is the chemical opposite of an acid, capable of neutralizing it to provide relief.
-
Explore the benefits of aluminum hydroxide as an antacid and its role in pharmaceuticals. ↩
-
Understanding antacids is crucial for safe consumption and effective relief from heartburn. ↩
-
Learn about the critical purification processes that ensure the safety of antacids. ↩
-
Learn about Magnesium Hydroxide’s unique properties and its use in antacid formulations. ↩
-
Understanding the pH scale is essential for grasping the concepts of acidity and basicity. ↩
-
Understanding heartburn can help you choose the right antacid for relief. ↩
You may also be interested in:

What protective measures should be taken when handling aluminum hydroxide powder in a factory?
Are you worried about worker safety when handling fine powders? Aluminum hydroxide is generally safe, but mishandling its dust can lead to serious respiratory issues and even create explosive conditions.

What Key Technical Indicators Should Be Considered When Purchasing Aluminum Hydroxide?
Are you struggling to find the right aluminum hydroxide for your needs? You might be paying for features you don’t even require. Let’s simplify the technical details. When buying aluminum

What are the market trends for aluminum hydroxide in 2026?
Are you planning your 2026 raw material purchases? Volatile markets make it hard. I will share insights from the factory floor to help you make better decisions for your business.

Is aluminum hydroxide toxic and is it harmful to the human body?
Worried about the word "aluminum" in your products? It sounds scary and can make you question its safety. I’m here to clear up the confusion with simple facts. Aluminum hydroxide

What are the side effects of long-term use of stomach medications containing aluminum hydroxide?
Struggling with constant heartburn? Reaching for that stomach medicine might seem like the only option. But what if that relief comes with hidden long-term costs? The most common side effect

Does ingesting aluminum (such as through aluminum hydroxide) cause Alzheimer's disease?
Are you worried that everyday products containing aluminum might be harmful? This fear connects to serious health concerns like Alzheimer’s, making you question what is safe. Let’s look closely. Based

Why do some vaccines contain aluminum hydroxide (aluminum adjuvant)?
Confused about vaccine ingredients? Seeing aluminum hydroxide listed can be unsettling. I’ll explain its crucial role and why it’s there to help your body build strong immunity. Aluminum hydroxide acts

Is the aluminum adjuvant in vaccines safe for infants or adults?
You read about vaccine ingredients and worry. The word "aluminum" sounds scary, especially when talking about babies. But understanding the facts can give you peace of mind. Yes, the aluminum

What environmental problems are generated during the production of aluminum hydroxide?
Worried about environmental compliance in your supply chain? Sourcing from China can be complex. You need a reliable, eco-conscious partner for your aluminum hydroxide needs. The main environmental issue is

Where are China's main aluminum hydroxide production areas?
Are you struggling to find the right aluminum hydroxide supplier in China? This confusion can lead to higher costs and unstable quality, which hurts your business and your reputation. China’s